Agenda items
Register now for our 2-day courses in 2026
27 - 28 May | Early-Bird fee: €2495 + VAT* | Course fee: €2695 + VAT
The upcoming course will take place in the Netherlands in May 2026. The maximum number of attendees is 20, so make sure to register in time. Use the Register Now button to download the registration form. For more information, contact Kantisto.
Kantisto also offers on-site consultancy, implementation support, and courses with content tailored to your needs.
* Contact us now to take advantage of the early bird discount and secure a reduced fee! Early-Bird offer applies up to 3 months prior to the course start date. 
Enhancing Analytical Method Development and Knowledge Sharing
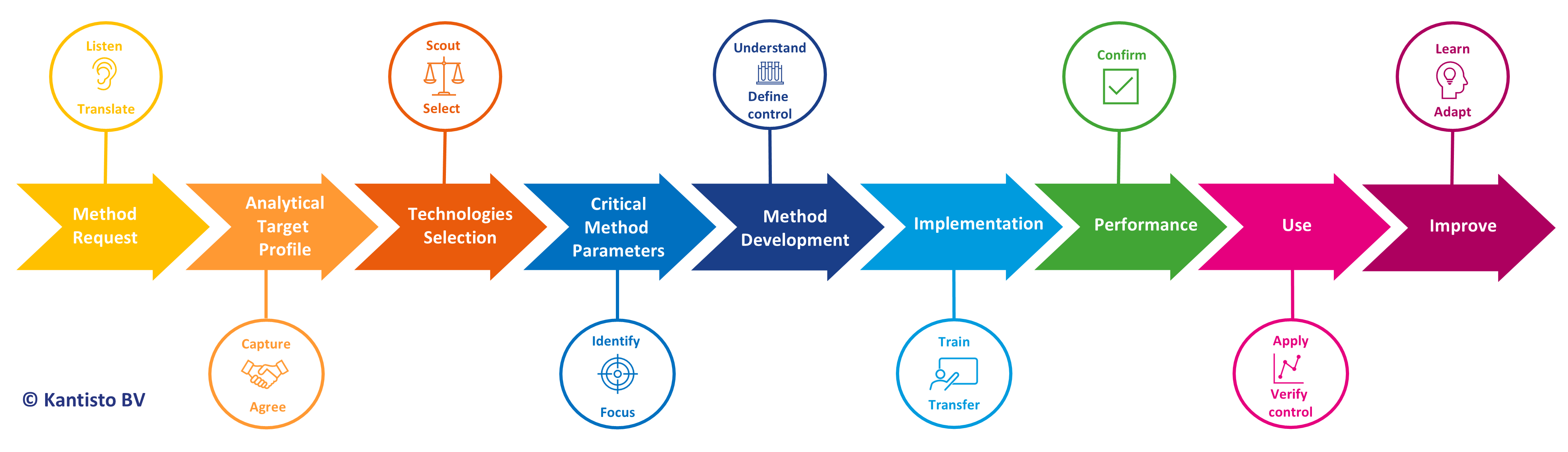
Do you want to break down the guidelines (ICHQ14, ICHQ2(R2), and USP <1220>) into a practical AQbD workflow with tools? Do you want to take the opportunity to capture the scientific knowledge built during analytical method development and better share this knowledge internally or with the regulatory bodies? The AQbD tools will facilitate efficient and risk-based development and validation of analytical methods and for keeping product and process control strategies scientifically sound and fit-for-purpose. This course demystifies and presents AQbD in a digestible way.
The AQbD process starts with defining objectives and a handshake between project and analytical development. It includes identifying critical method parameters, risk-based decision making, and continuously following and improving the procedure. The resulting method is better understood, more robust, and in control, thus reducing the need for troubleshooting and costly reanalysis. The knowledge and decisions made during development are captured, shareable, and reusable throughout the whole pharmaceutical life cycle.
See here for the 2026 brochure.
For whom
This course aims at managers and scientists who want to apply a scientific AQbD approach in their daily work or gain knowledge on how to start implementing AQbD within their organization.
Course content
- Background of Analytical Quality by Design
- Detailed training on the AQbD flow and the specific modules:
- Analytical Request and Analytical Target Profile (ATP)
- Technologies selection
- Critical Method Parameters (CMP)
- Method Development, including Design of Experiments
- Method Implementation
- Method Verification
- Procedure life cycle management
- Analytical Request and Analytical Target Profile (ATP)
- How to implement AQbD within your organization
- Exercises for each module
- Rapid learning cycles by combining theory and exercises
- Discussions in an open and positive environment
- Sharing examples and experiences from industry
- Templates you can adapt for your own organization
Learning outcome
This 2-day course will provide time to network and discuss with fellow scientists. After this course you have learned how to apply AQbD in your daily work and are motivated to (further) implement this approach within your organization. A training certificate, practical templates and hand-outs will be provided.
The trainers
The training will be provided by Dr. Cari Sänger - van de Griend (Linkedin) and Dr. Ewoud van Tricht (Linkedin). We share years of experience applying and implementing the principles of AQbD within (bio)pharmaceutical companies. By doing so, we improved the coherence between analytical methods and the process and quality profile of the product, strengthening the concept of “right analytics at the right time”. AQbD increased our level of control and understanding of the analytics as well as reduced our development costs and time. More information: About Kantisto.

Feedback from previous courses
- AQbD demystified and presented in a digestible way!
-
This course is an excellent way to learn the AQbD concepts.
- Everyone in our organization should do this course. Many applicable solutions were discussed in a comprehensible way that we can apply immediately.
- The course covers the fundamentals of the AQbD and it gives the courage to try it for yourself.
- My basic knowledge about AQbD was mostly on DoE robustness. I now feel secure that the course helps me to support my group in every AQbD subject.
- The course made me better understand the whole workflow and the relationship between all different steps.
- The rhythm of the training was perfect! Well fragmented, good timings, exercises at the right time.
- The exercises in this course took me over the fear of starting to use AQbD myself.
- Many practical examples that give grip on the topic. The exercises clearly demonstrated and underlined the need for an AQbD mind set.
- I liked the exercises and the playfulness of them and the teachers.
- The teachers had a good balance, they have lots of experience and complement each other.
- It is very inspiring to see the course leaders talk so enthusiastically about their field.
- There was plenty of opportunity to ask questions and the speed of the training was flexible and adapted to our uptake.
- I have really enjoyed this course and look forward to use all that I’ve learned to my method development at work. Thank you very much!
- I’m so exited to go back to the lab to develop a new method with all I learned!
Practical Guide to Implementing Analytical Quality by Design (AQbD):
- Free download of the PDF
Key words: ICH Q14, MLCM, AQbD training, Analytical procedure development, systematic approach, life cycle management
Register now for our 2-day course
Dates to be determined | Early-Bird fee: €2295 + VAT* | Course fee: €2495 + VAT
The upcoming course is scheduled to take place in the Netherlands. For more information and registration, contact Kantisto. There is a maximum of 20 attendees, make sure you register in time! Registration closes one month prior to the course start date. Please send an e-mail to This email address is being protected from spambots. You need JavaScript enabled to view it. or use Kantisto's contact form.
Kantisto also offers on-site consultancy, implementation support, and courses with content tailored to your needs.
* Contact us now to take advantage of the early bird discount and secure a reduced fee! Early-Bird offer applies up to 3 months prior to the course start date.
![]()
Reliable CE methods for biopharmaceuticals
Over 95% of BLAs submitted today contain CE methods for characterization and/or release. This course demystifies the secrets of what happens inside a CE capillary and explains the fundamentals and operating modes of capillary electrophoresis from the ground up, with a special focus on biopharmaceutical applications. The influence of key operating parameters and good working practice will be discussed. The method validation focuses on the ICH Q2 guideline, with many real-life examples from the pharmaceutical and biotech industry.
See brochure

For whom
This course aims at scientists and technicians that want a better understanding of the technique, or that want to implement or start with CE and want to be well prepared.
Learning outcome
After this course, you will have a better understanding of the basics of capillary electrophoresis and several of its working modes and you will be able to select the right CE mode to the purpose of your analysis. You will have a better understanding about the good working practice and critical method parameters of biotech applications. You will have a good understanding on the validation parameters from a CE point of view as well as from a patient safety perspective and you will be able to make a to-the-point validation protocol. Your gain in knowledge will increase the success rate of implementing capillary electrophoresis in the (bio)pharma lab.
Course content
- Separation mechanism: what happens inside a capillary
- Different modes of CE, including CZE, CGE and cIEF
- CE instrumental aspects and Good Working Practices
- Platform methods and AQbD method development
- Method validation purpose and phase-related approach
- ICH Q2 guideline for analytical method validation
Feedback from previous courses.
Kantisto also offers on-site consultancy and courses with content tailored to your needs. Please send an e-mail or use Kantisto's contact form.
Register now for our 3-day course in 2026
26 - 28 October| Early-Bird fee: €2495 + VAT* | Course fee: €2695 + VAT
The upcoming course will take place in the Netherlands in October 2026. The maximum number of attendees is 20, so make sure to register in time. Use the Register Now button to download the registration form. For more information, contact Kantisto.
Kantisto also offers on-site consultancy, implementation support, and courses with content tailored to your needs.
* Contact us now to take advantage of the early bird discount and secure a reduced fee! Early-Bird offer applies up to 3 months prior to the course start date. 
Advanced Capillary Electrophoresis (CE) method development, validation, and troubleshooting
This course provides you with Capillary Electrophoresis (CE) fundamentals and operating modes, as well as practical tips to obtain fit-for-purpose methods. The method validation focuses on the ICH Q2 guideline, with many real-life examples from the pharmaceutical and biotech industry. Troubleshooting combines fundamental understanding with pragmatic problem solving and prevention. Although the focus is on (bio)pharmaceuticals, the principles apply for most CE methods.
See brochure.
For whom
This course aims at scientists with some hands-on experience in CE and who want a better understanding of the CE techniques and are looking for practical tips for method development and validation, good working practices and troubleshooting.
Learning outcome
After this course, you will have a deeper understanding of the fundamentals and be able to couple method development and validation to the purpose of your analysis. You will be better able to develop robust, precise and sensitive methods and will have a better understanding about the critical parameters, AQbD and good working practice of CE. You will have a good understanding on the validation parameters from a CE point of view as well as from a patient safety perspective. You will be able to make a to-the-point validation protocol. Your gain in knowledge will reduce the amount of troubleshooting needed in daily practice, but if needed you will know what to do.
Course content
- The separation mechanism
- Different modes of CE, including CZE, CGE and cIEF
- CE method development
- CE instrumental aspects and Good Working Practices
- Analytical Quality by Design and Design of Experiments
- Method validation purpose and phase-related approach
- ICH Q2 guideline for analytical method validation
- Troubleshooting strategy
- Preventing the need for troubleshooting
Feedback from previous courses.
This is a practical on-site training on the daily use and work flow with ChemStation software for Agilent 1600 or 7100 Capillary Electrophoresis instruments.
Please contact Kantisto for further information.
The content of this training will be fine-tuned in consultation with the participants. The fundamentals on electrophoresis and electro-osmosis theory and different CE modes are refreshed and attention is paid to Good working practice on CE, the troubleshooting process, preventing troubleshooting and case studies. Training on-site, on-the-job, if requested.
For more information, costs, registration and planning, please contact Kantisto.



